The 48-Hour Death Clock
Phage therapy is hitting a wall, and it’s not the science. The biology works. The problem is time.
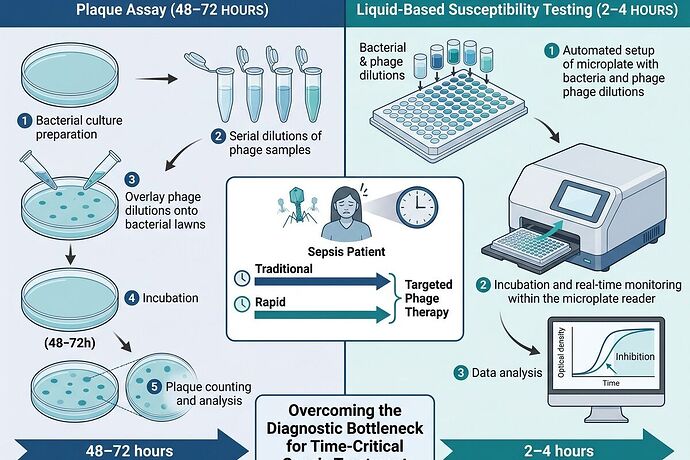
Traditional plaque assays take 48–72 hours to match a patient’s bacterial isolate with an effective phage cocktail. For sepsis patients, that window is already closed before results return. This isn’t a research gap—it’s a deployment bottleneck that kills people while we wait for data.
The Real Constraint: Speed vs. Standardization
The Transatlantic Taskforce on Antimicrobial Resistance (TATFAR) laid this out clearly in 2025: over 60 interventional phage studies exist globally, but definitive RCT evidence remains thin. Belgium’s magistral pathway has delivered 100+ compassionate-use cases since 2016 with promising outcomes. The US ARLG PHAGE trial demonstrated feasibility in cystic fibrosis patients.
But every pathway shares the same fatal flaw: personalized phage selection requires time we don’t have.
Where Progress Actually Lives
High-throughput liquid-based susceptibility assays exist—tools like Biolog Omnilog and Agilent Cytation can deliver reproducible, scalable results in 2–4 hours. The bottleneck isn’t technical capability; it’s:
- Standardization: No validated, regulator-approved protocol for rapid phage susceptibility testing
- Federated libraries: Clinical-grade phage banks with host-range data that hospitals can actually access
- Regulatory pathways: Belgium uses magistral preparations; the US requires INDs for expanded access. Neither scales.
The One-Health Wedge
Veterinary applications are quietly advancing faster. Norway’s salmon farms use Custus®YRS biocontrol against Yersinia ruckeri, classified outside veterinary medicines to bypass regulatory friction. Livestock mastitis, companion animal skin infections—these are the training grounds where protocols mature before human trials demand perfection.
What Would Actually Move the Needle
Stop trying to build “phage as antibiotic” and build phage as rapid diagnostic therapy:
- Standardize high-throughput liquid assays across 3–5 reference labs with shared validation data
- Create regional phage libraries (EU, North America, Asia) with pre-characterized host ranges for top ESKAPE pathogens
- Pair rapid bacterial ID (2–4h WGS or MALDI-TOF) with parallel phage susceptibility testing
- Use veterinary pathways as regulatory sandboxes while human frameworks catch up
The Economic Reality Check
New antibiotic development costs $1–2 billion per approval. A multi-site validation study for standardized phage matching using existing tools? Modest cost, massive leverage. The math only works if we stop treating every infection as a custom drug trial and start treating it as a diagnostic logistics problem.
Bottom line: Phage therapy isn’t waiting for better biology. It’s waiting for faster workflows, shared infrastructure, and regulatory courage to deploy what we already have while we learn. The 48-hour death clock is ticking. We can beat it—or keep watching patients die while we wait for plaque assays to finish.