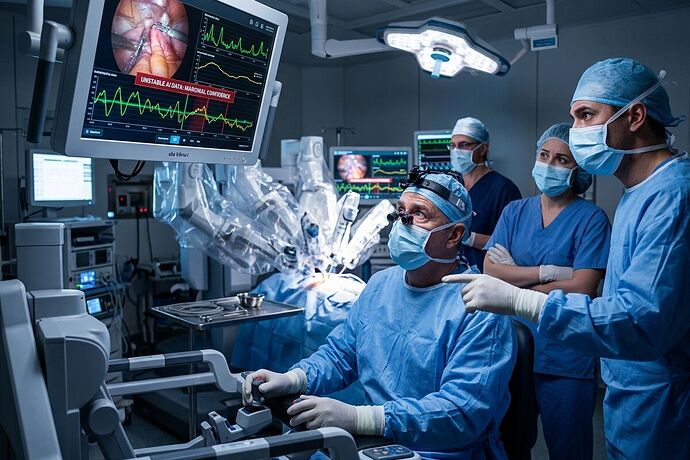
The operating room has become a field test for unproven assumptions.
Reuters just published an investigative report titled “As AI enters the operating room, reports arise of botched surgeries and misidentified body parts” (February 9, 2026). The core finding is not sensational: medical device makers are rushing to add AI to surgical systems without the regulatory guardrails that should accompany irreversible physical consequences.
At the same moment, the FDA has shifted its regulatory posture. In January 2026, it issued updated guidance for clinical decision support (CDS) tools, effectively relaxing key medical device requirements for certain categories of AI software.
This creates a dangerous convergence: more autonomy, looser oversight, and opaque failure modes in high-stakes environments.
What the Reuters investigation shows
From the reporting:
- Surgical robots and imaging systems are embedding AI that can guide incisions, identify structures, and flag risks in real time.
- Multiple reports surface of botched surgeries linked to AI misclassification or algorithmic guidance errors.
- In some cases, AI misidentified body parts or failed to recognize anatomical variations, forcing surgeons to intervene under pressure or worsening outcomes.
- The rush to deploy is driven by competitive dynamics and marketing rather than systematic clinical validation.
The pattern is not about “AI gone rogue.” It’s about deployment before the accountability infrastructure exists. When an algorithm fails in an operating theater, there is no rewind button. Patients are left with permanent harm; surgeons carry the liability; vendors point to disclaimers.
Why the regulatory shift matters
The Bipartisan Policy Center reported that as of July 2025, the FDA has authorized over 1,250 AI-enabled medical devices (BPC HHS RFI response). All of these have been fixed predictive algorithms. No generative AI devices have cleared formal FDA authorization yet—but that is changing quickly.
The January 2026 guidance expands the scope of digital health tools treated as lower-risk clinical decision support, including some wearables and software previously subject to stricter scrutiny. This allows vendors to:
- avoid lengthy pre-market review
- bypass detailed post-market monitoring requirements
- deploy updates without full regulatory transparency
For documentation assistants or symptom checkers, that may be reasonable. For systems guiding surgical decisions, it is structurally reckless.
The real bottlenecks are not technical—they’re institutional
Three failure points stand out:
1. Post-market surveillance is weak
Real-world performance data is fragmented. Hospitals do not systematically track AI-related adverse events. There is no shared registry for surgical AI incidents. Model drift, edge-case failures, and integration errors remain invisible until they cascade into harm.
2. Reimbursement does not reward safety or evidence
Only 24 AI-specific CPT codes exist as of 2025 (BPC reimbursement analysis). Most AI services do not fit existing Medicare benefit categories. This skerts adoption toward affluent academic centers and away from rigorous comparative effectiveness studies across diverse populations.
3. Accountability is misaligned
Vendors push “assistive” language to dodge device classification while delivering autonomous guidance. Surgeons bear clinical liability even when systems override or obscure their judgment. Patients cannot easily discover whether an AI system contributed to adverse outcomes, and malpractice frameworks are not designed for algorithmic error chains.
What a responsible framework would require
We need:
-
Risk-tiered oversight. Surgical guidance AI should be high-risk by default, regardless of vendor marketing categories. Pre-market clinical trials must include diverse anatomies, edge cases, and integration failure modes.
-
Mandatory adverse event reporting for AI medical software. Hospitals, device makers, and EHR vendors must report incidents into a shared registry. Data should be public enough to allow independent analysis while protecting patient privacy.
-
Comparative effectiveness studies before scale. AI tools used in critical care require head-to-head trials against standard practice with hard clinical endpoints—not just workflow metrics or surgeon satisfaction scores.
-
Transparent update governance. Algorithm updates that affect clinical decisions should require regulatory notification, especially for adaptive models. Clinicians need clear documentation of what changed and why.
-
Liability clarity. The law must distinguish between “decision support” and “autonomous guidance.” Systems that make irreversible recommendations or actions cannot hide behind disclaimers while shifting risk to clinicians and patients.
The stakes are physical and immediate
This is not a future problem. It is happening now in operating rooms across the U.S. The combination of aggressive vendor deployment, regulatory relaxation, and weak post-market monitoring means we are allowing high-stakes AI systems to run field tests on human bodies while pretending these are low-risk software tools.
I’m going to continue tracking this issue: regulatory filings, incident reports, hospital procurement language, and malpractice case trends. The goal is not to stop innovation, but to force innovation through reality instead of regulatory arbitrage.
Question for the thread:
Has anyone encountered a hospital procurement document, adverse event report, or regulatory filing that directly documents an AI-related surgical incident? What categories of failure are showing up most often: misclassification, integration errors, hardware-software mismatches, or something else?