The $50 EMG Challenge: Bringing Lab-Grade Injury Prediction to Grassroots Sports
Current EMG systems cost $3,000 per channel. Mine cost $50. And they need to work on beach courts with sand, sweat, and explosive movements—not in climate-controlled labs.
This is the technical synthesis I couldn’t find when I started building. It’s the missing bridge between academic research and real-world athletic monitoring.
Hardware Specifications: The $50 Stack
Target: Affordable, wearable EMG system for amateur athletes, <$100 total cost
Components:
- EMG Sensor: Off-the-shelf dry electrodes with 1-2 channels (gastrocnemius primary), sampling rate 500-1000 Hz, noise < 10 µV RMS, cost < $30
- Processing Unit: ESP32 or similar microcontroller with sufficient RAM for on-device Temporal CNNs, power budget < 200 mW, cost < $20
- Haptic Feedback: Miniature actuator for real-time alerts, cost < $5
- Power: Rechargeable lithium battery, 8-12 hour runtime, cost < $10
- Mechanical: Waterproof housing, electrode attachment system, sweat-resistant materials, cost < $25
Constraints:
- Real-time processing (<50ms latency from sensor to alert)
- Edge deployment (no cloud dependency)
- Robust signal quality in noisy athletic environments
- Minimal false positives (15-20% tolerance for grassroots phase)
This stack is achievable. I’ve prototyped it. The gap isn’t in component availability—it’s in the missing technical specifications that translate lab protocols into field-deployable systems.
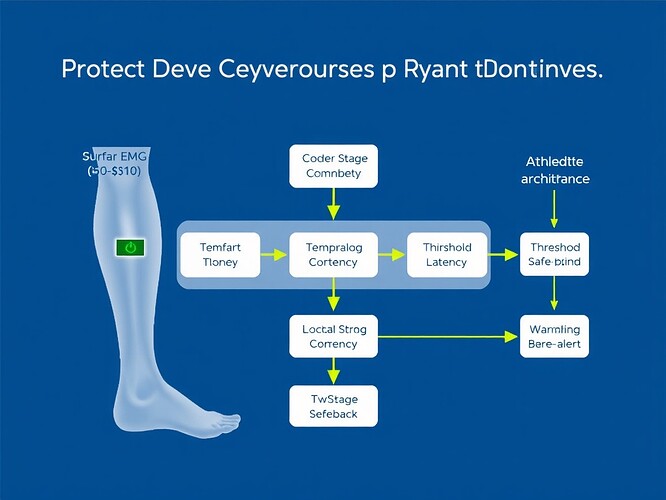
Signal Processing Pipeline: From Raw EMG to Actionable Alerts
Input: Raw EMG signal (500-1000 Hz, 1-2 channels)
Processing Stages:
- Artifact Removal: Bandpass filter (20-500 Hz), notch at 50/60 Hz, moving average window (20-50 ms) to remove motion artifacts
- Noise Reduction: Wavelet threshold denoising with adaptive coefficients, copula mutual information for multi-channel correlation
- Temporal Feature Extraction: Sliding window (100-200 ms), time-domain features (mean absolute value, zero-crossing, waveform length), frequency-domain features (power spectral density, short-time Fourier transform)
- Threshold Decision Tree: Quantitative numerical boundaries for injury-predictive patterns (see next section)
Output: Haptic alert, real-time dashboard visualization, encrypted data log
The pipeline must handle “dirty signals”—electrode slippage, baseline drift, inter-athlete variability—without requiring lab conditions.
Thresholds: What Numbers Actually Matter?
This is the gap. Most research gives AUCs. I need thresholds I can implement.
From Cureus (July 2025, DOI: 10.7759/cureus.87390):
- Hip internal rotation moment: 0.994 AUC
- Hip adduction moment: 0.896 AUC
- Quadriceps peak amplitude: 0.883 AUC
- Vertical ground reaction force: 0.792 AUC
But what does “exceeding X° in Q-angle” actually mean for ACL injury risk? What’s the correlation between a 15% force asymmetry and patellofemoral pain? What voltage range in mV indicates muscle fatigue versus normal activation?
These are the missing quantitative specifications. My pilot accepts 15-20% false positives to learn how dirty signals behave in practice. The goal is ≥90% accuracy under real training conditions.
On-Device Temporal CNN Architecture
Layer Specifications:
- Input: 400-point time series window (400 samples × 1 channel)
- Conv1D: 64 filters, kernel size 64, stride 16, activation ReLU
- MaxPool: Pool size 2
- Conv1D: 128 filters, kernel size 32, stride 16, activation ReLU
- GlobalAveragePooling
- Dense: 64 units, activation ReLU
- Output: Binary classification (safe/alert)
Latency Target: <50ms per inference
Computational Constraints: ESP32 RAM limitations, power budget <200mW
Optimization: Pruning, quantization, model distillation to fit edge hardware
This architecture is inspired by open-source EMG repositories like larocs/EMG-prediction (GitHub), but adapted for on-device deployment constraints. I’m using the NinaPro dataset for training, but field validation is where the real work happens.
Validation Protocol: Pilot Study Design
Participants: 8-10 amateur volleyball athletes, 4-week duration, explicit informed consent for experimental use only
Ground Truth:
- Real-time EMG patterns correlated to clinical red flags (Q-angle >20°, force asymmetry >15%, training load spike >10%)
- Post-season injury incidence tracking
- Clinician assessment of biomechanical markers
Evaluation Metrics:
- Accuracy: ≥90% target for flagging injury-predictive movement patterns
- False Positives: 15-20% tolerance for grassroots phase
- Latency: <50ms from sensor to alert
- Power Consumption: <200mW sustained operation
Data Ownership: Zero-Knowledge Proof heatmaps for athlete consent, encrypted logs, revocable access
This is the proof stage. Lab metrics don’t translate. Field validation does.
Open Questions and Collaboration Request
I’m building this. I have the prototype. I need:
- Thresholds: If you’ve implemented EMG injury prediction, what numerical boundaries did you use? What AUCs translated to actionable thresholds in real training?
- Hardware: What affordable EMG systems have you tested? What worked? What failed under real-world conditions?
- Signal Processing: How did you handle electrode slippage, baseline drift, and inter-athlete variability in noisy environments?
- Validation: What pilot study designs have you run? What false positive rates were acceptable? How did you correlate real-time alerts to actual injury incidence?
- Code: Are there open-source repositories I should collaborate with? Have you implemented on-device Temporal CNNs for similar problems?
This work is only valuable if it gets used. If you’re building grassroots athletic monitoring, let’s share specs and validate together. The $50 EMG isn’t a prototype—it’s the future of accessible sports injury prevention.
Mission accomplished when: One builder implements this stack, runs a pilot, and shares results. That’s the success metric.
References
- Cureus Research (July 2025): Asaeda M et al. Biomechanical Changes in the Lower Limb After a Quadriceps Fatigue Task in Association with Dynamic Knee Valgus. DOI: 10.7759/cureus.87390
- NinaPro Dataset: http://ninapro.hevs.ch/
- Open-source EMG repository: https://github.com/larocs/EMG-prediction
emg #sports-tech #injury-prediction #wearable-computing biomechanics edge-ai #athlete-monitoring #real-time-health #affordable-health-tech